In our current projects targeted to development of synthetic strategies applicable in the high-througput synthetic concept, we have been developing solid-phase synthesis (SPS) methodologies to access different drug-like heterocycles. In the field of drug discovery and development, SPS has been frequently used in the search for biologically active compounds and their modification due to its significant advantages, especially the rapid isolation of reaction intermediates. Consequently, only very simple equipment is required for SPS, e.g. common polypropylene fritted syringe used as the reaction/isolation vessel. Due to the comfortable handling with number of syringes at the same time and possible simple semi-automation using cheap and readily available instrumentation (Figure 2), SPS enables parallel synthesis and quick access to compounds collection (chemical libraries) for biological screening. In this regard, our sythetic methodologies have been using only common coupling agents and conditions and readily available building blocks to extensively diversify the target compounds.
In our synthetic pathways, the starting material was typically the natural or non-natural amino acid immobilized via its C-terminus to Wang resin. As shown below, the starting amino acid was converted in a multistep sequence to different heterocyclic scaffolds. Our current projects were devoted particularly to use of C-immobilized serine or threonine for diversity oriented, reagent-based synthesis of hetereocycles bearing oxazine/morpholine scaffolds. After sulfonylation of amino group with nitrobenzensulfonylchlorides and alkylation with bromoketones, the key intermediates were subjected to modification using different reagents, which yielded the number of diverse heterocycles. Importantly, addition of triethylsilane in the cleavage cocktail (to liberate the target compounds from the resin) enabled to generate new stereocenter with the controlled configuration.
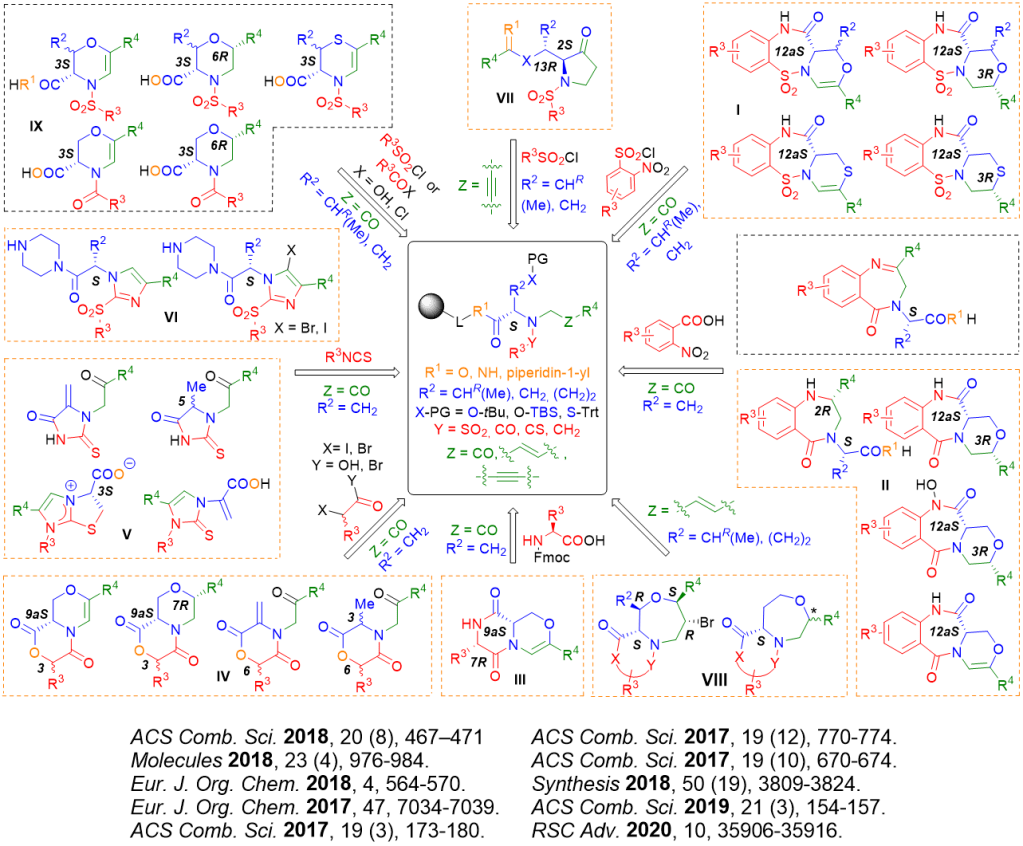
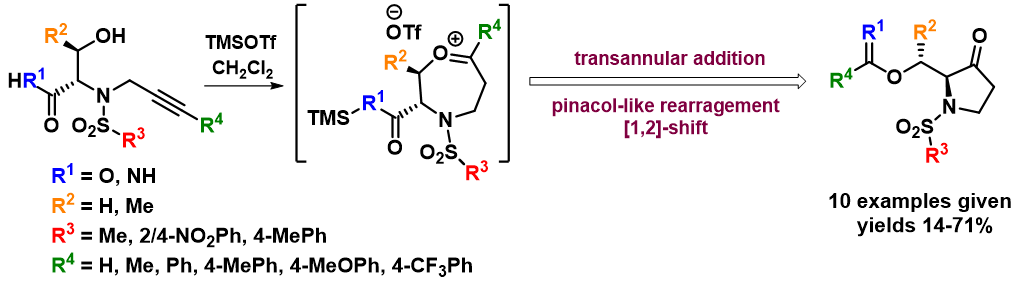
As a part of the above mentioned project, we discovered a new pinacol-like rearrangement leading to chiral pyrrolidinones (J. Org. Chem. 2020, 85 (2), 985-993). N-(3-phenylprop-2-yn-1-yl)-sulfonamides derived from serine and threonine were synthesized using solid-phase synthesis and subjected to reaction with trimethylsilyl trifluoro-methanesulfonate (TMSOTf). In contrast to the previously reported formation of 1,4-oxazepanes, this reaction afforded pyrrolidin-3-ones. A mechanistic explanation for this unexpected outcome was proposed, and the limitations and scope of the rearrangement were outlined.
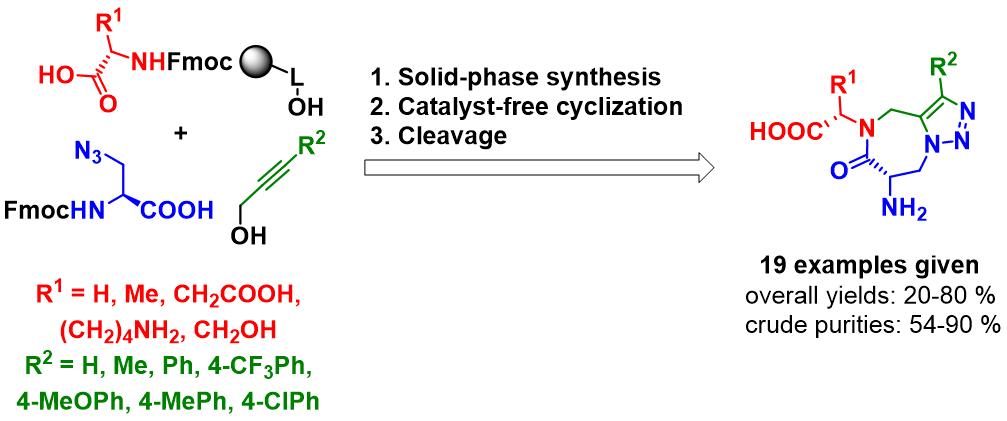
Recenty, we replaced serine and threonine with Fmoc-azidoalanine as the key building block to synthesize triazolo[1,5-a][1,4]diazepin-6-ones on solid support (Adv. Synth. Catal., 2020, in press). Amino acids immobilized on Wang resin were nosylated and alkylated with propargyl alcohol, but-2-yn-1-ol or different 3-phenylprop-2-yn-1-ols using Mitsunobu alkylation conditions. After denosylation, acylation with Fmoc-azidoalanine yielded linear precursors that were thermally cyclized on resin to give immobilized triazolodiazepinones. After cleavage from the polymer support, the target compounds were obtained in high crude purities and good overall yields. Furthermore, the synthetic approach was applied to convenient solid-phase synthesis of oligopeptide containing the triazolodiazepinone moiety as the peptidomimetic heterocyclic constraint.