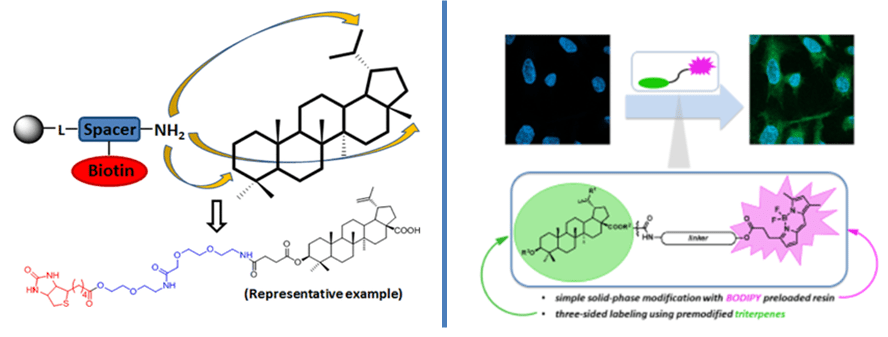
In this field, we have been focusing on the development of synthetic tools to enable the simple preparation of different bioconjugates for chemical biology. The general strategy applied preparation of preloaded resin and the use of the „shake-wash-cleave“ procedure. Our first reported preloaded resin yielded conjugates of semisynthetic triterpenes with cytotoxic activity but unknown mode of action. Their conjugation with widely used affinity tag biotin led to compounds applicable for biotin-streptavidin interaction assays (Bioconjug. Chem. 2015, 26, 2563). In the following years, we focused on visualization of small molecules in cells which becomes an essential tool in drug discovery. The most commonly used method is fluorescent microscopy, where a studied molecule is fluorescently labeled and directly visualized within the cell. A number of various conjugates of small molecules with fluorescent tags were reported to date with the application as probes, photosensitizers, or luminescence switches and sensors. We focused on the development and study of conjugates of cytotoxic triterpenes with BODIPY (Chem. Eur. J. 2018, 24, 4957).
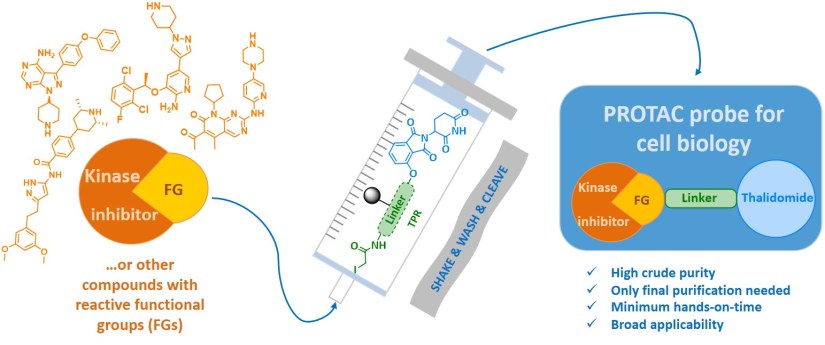
The approach of preloaded resins was also used in the field of proteolysis targeting chimeras (PROTACs). PROTAC conjugates consist of two main parts connected through a suitable linker, where one part interacts with the protein of interest, whereas the other binds to a component of E3 ubiquitin ligase. The phthalimide family is often employed as an E3 ligase binder to hijack cereblon to target proteins. Pharmacological targeting of protein kinases has been validated as an effective therapeutic strategy, and over 37 kinase inhibitor drugs have received approval for clinical use in certain cancers. However, specific resistance often reduces the sensitivity of targeted kinases to drugs during therapy, and therefore novel molecules or approaches are intensively sought. Kinase degradation induced by PROTACs represents an interesting alternative not only for mechanistic studies or therapy but also for kinases with kinase-independent functions. In our contribution, we introduced a preloaded resin for high-throughput synthesis of PROTACs and degradation of different protein kinases (Chem. Commun. 2019, 55, 929).
Recently we prepared PROTAC conjugates designed for simultaneous proteolysis of two enzymes: FLT3 and CDK9. It shows selectivity towards FLT3-ITD mutated AML cells and its mechanistic study confirmed the effective degradation of target proteins (Eur. J. Med. Chem. 2022, 243, 114792).

The solid-phase synthesis was also used for the preparation of folic acid conjugates, although in this case without using the concept of preloaded resin (J. Org. Chem. 2017, 82, 13530). Little or no specificity to cancer cells is one of the most serious problems of traditional chemotherapy for cancer as it leads to systemic toxicity. The systemic toxicity of conventional chemotherapy causes serious side effects and is also one of the limiting factors of treatment efficiency. The use of small molecule delivery systems is a promising approach that can decrease the systemic toxicity of cytotoxic drugs to rapidly dividing normal cells. Tumor-targeting delivery systems have been intensively studied in the last three decades and represent one of the promising approaches for increasing chemotherapy selectivity together with decreasing systemic toxicity. Selective drug delivery is based on frequent overexpression of many receptors in tumor cells, which can serve as targets to deliver cytotoxic agents selectively into tumors. The relatively selective transport into tumor cells can be realized with the use of conjugates of cytotoxic agents and tumor recognition moiety.
Our research also focused on the synthesis of conjugates applicable for molecular imaging using positron emission tomography (PET). Our initial study aimed to synthesize deferoxamine-based c(RGD) peptide conjugate for radiolabelling with gallium-68 and perform its basic preclinical characterization including testing of its tumor-imaging potential. We have shown here that [68Ga]Ga-DFO-c(RGDyK) can be used for αvβ3 integrin targeting, allowing imaging of tumor-induced angiogenesis by positron emission tomography (Int. J. Mol. Sci. 2021, 22, 7391).